Introduction
As healthcare costs increase because of the aging population and technological developments in healthcare, the need by healthcare decision makers for economic evaluations of new healthcare interventions becomes more important. A comprehensive economic evaluation of a new healthcare intervention requires an analysis of both the efficiency of the intervention compared with current treatment patterns and the annual budget impact of the new intervention. An analysis of the annual budget impact might be used to determine affordability of the new intervention, given healthcare budget constraints, or as an implementation tool for newly reimbursed interventions.
A budget-impact analysis typically first identifies, in a national or local health plan, the treated population for the indication for which the new intervention is approved. The analysis then estimates the annual change in healthcare expenditures for the treated population with and without the new intervention in the treatment mix for different rates of uptake of the new intervention. Unlike a cost-effectiveness analysis, which compares the new intervention with a standard of care, the comparison in a budget-impact analysis is between the mix of treatments before the new intervention is reimbursed and the mix of treatments after the new intervention is reimbursed, taking into account the rate of uptake of the new intervention.
There are several published guidelines for budget-impact analyses. These guidelines have been developed either by the health technology assessment (HTA) agencies that require a budget-impact analysis as part of a reimbursement submission (e.g., Pharmaceutical Benefits Advisory Committee (Australia), Canada, Taiwan, and the National Institute for Health and Clinical Excellence (NICE) in the UK) or by independent organizations (e.g., the International Society for Pharmacoeconomics and Outcomes Research). These guidelines describe the estimation framework and data sources that are recommended for performing budget-impact analyses.
Key Elements Of A Budget-Impact Analysis
Budget-impact analyses have six primary elements, irrespective of the modeling framework used to derive the estimates: (1) treated population size, (2) time horizon, (3) treatment mix, (4) intervention costs, (5) other healthcare costs, and (6) presentation of results. In addition to these six primary elements, budget-impact analyses generally include sensitivity analyses to test the impact on budget estimates of the uncertainty in the input values used in the analysis or the variability of these inputs among health plans or health systems. Issues that should be considered for each of these elements are described in the following paragraphs.
The first step in a budget-impact analysis is to determine the population currently being treated for the disease indication of interest using epidemiological data. It is critical to estimate not only the size of the treated population but also the mix of disease severity in the population because treatments and disease-related healthcare expenditures may vary with disease severity. For example, individuals with schizophrenia that is refractory to treatment with standard care will have higher annual costs and will use a different mix of treatments than individuals who are responsive to treatment. It also is important to consider a possible ‘woodwork’ effect with a new intervention, that is, more patients with the indicated condition presenting for treatment when a better treatment becomes available. Finally, for a new intervention that reduces mortality, slows disease progression, and/or changes treatment patterns, changes in the treated population size and the distribution of the population by disease severity must be estimated on the basis of the assumed uptake rates for the new intervention.
The second element, the time horizon for the budgetimpact analysis, typically is chosen on the basis of the requirements of the healthcare decision maker, rather than on the duration of the impact of the new treatment (as for a cost-effectiveness analysis). Because healthcare budget holders generally have a short planning horizon, time horizons of 3–5 years are usual. With such short time horizons, offsetting cost savings many years in the future from slowed disease progression of chronic diseases or prevention of future cases of the disease or its complications are not captured. But this is an accurate reflection of the costs incurred over the typical planning horizon.
The third element in a budget-impact analysis is the determination of the mix of interventions currently used for the indication and the predicted change in that mix if the new intervention is made available. Unlike cost-effectiveness analyses, which compare the outcomes when taking the new intervention with the outcomes with a standard-of-care intervention, a budget-impact analysis does not assume immediate switch by all patients to the new intervention. Rather, the new intervention is assumed to alter the mix of interventions used for the indication, using estimated or observed uptake data. The budget impact will be higher if the new intervention is used in place of a generic drug than if the new intervention is used in place of another branded drug or a surgical procedure. Also, the budget impact will be higher if the new intervention is combined with current treatments instead of substituted for them.
The costs associated with the current and new interventions should include some or all of the following, depending on the type of intervention: acquisition, administration or labor, other equipment, monitoring, and adverseevent or complication costs. For drugs, generally, wholesale acquisition costs (in the US) or national formulary costs are used as the default values, although the analysis should be designed so that discounts and copays can be subtracted from these costs to provide more accurate estimates of the healthcare decision makers’ costs. For devices, wholesale prices should be used; for procedures, standard labor costs should be used. All of these costs are used to reflect the expected costs of current and new interventions to the decision maker for each year of the budget-impact analysis time horizon.
The fifth element, an estimate of the impact of the new intervention on other indication-related costs, excluding intervention costs, is generally but not always included in budget-impact analyses. A simple calculation can be used, based on clinical trial data, for example, to estimate these costs for acute conditions and for those chronic conditions where the full impact on indication-related costs happens within a short period of time or is not likely to change over the model time horizon. Alternatively, changes in indication-related costs for a chronic illness may be estimated by adapting the disease progression model (used to estimate the cost-effectiveness ratios) to calculate annual indication-related costs after reimbursement has been approved for the new treatment. This adaptation involves running the cost-effectiveness model in ‘prevalence’ mode where the model adds a newly treated cohort each subsequent year, in addition to tracking the starting cohort.
The sixth element in budget-impact analysis is the presentation of the results. Unlike cost-effectiveness analysis, where there may be a societal perspective that can be used as the Bibliography: case, there is no Bibliography: case in budget-impact analysis. The appropriate perspective for the analysis varies with each decision maker’s budget responsibilities, which may range from a pharmacy or department budget to an entire hospital or outpatient clinic budget to countrywide healthcare services. Thus, the model needs to be programed in such a way that it can generate the budget impact from these multiple perspectives. In general, the results are presented undiscounted for year 1, 2, 3, etc. after the new intervention is made available to the decision maker’s population. Cumulative, multiyear results also may be presented either discounted or undiscounted.
Clearly, in any budget-impact analysis, there is uncertainty about both model assumptions and input parameter values. In cost-effectiveness analysis, one-way and probabilistic sensitivity analyses are the recommended approaches for presenting the impact of the input parameter uncertainty. For budget-impact analyses, the more common approach to uncertainty analysis is to present a series of scenario analyses, changing input parameter values either one at a time or several at a time to create different scenarios that are meaningful to the decision maker; for example, changing intervention uptake rates and/or expected effectiveness in the decision maker’s population. The decision maker may also enter values for input parameters that may vary among health plans or health systems but be known with certainty to each decision maker, such as drug costs, treated population size (based on size of population served and local incidence or prevalence rates), disease severity mix, and patient age distributions. Scenario analyses, which include alternate combinations of uncertain and variable input parameters, provide decision makers with more credible information about the range of possible results, given the specifics of their health plan or health system.
Categorization Of Budget-Impact Modeling Approaches
There are three main budget-impact modeling approaches that have been used by HTA agencies and/or in published studies: (1) cost calculator, (2) Markov or state transition model, and (3) Monte-Carlo or discrete-event simulation model. The simplest approach, a cost calculator, is typically used for acute indications and for chronic indications where a static analysis is appropriate; Markov models and discrete-event simulation models are used for chronic indications where a dynamic approach is needed to capture the changes in treated population size, indication severity mix, or treatment patterns.
Budget-Impact Analysis: Cost Calculator Approach
For each drug recommended for reimbursement by the National Health Service (NHS) in England, NICE prepares a costing template for the drug’s recommended use where budget impact is assessed to be greater than d1 million or more than 300 patients are affected. The costing template is presented on the NICE web site as a guide to budget planning for decision makers implementing the recommendation in the UK. These costing templates provide excellent examples of static models using a cost calculator approach. The NICE costing templates estimate the expected impact on the NHS budget of the new drug’s predicted market uptake over the next 3–5 years, after considering the current and new drug acquisition costs and associated administration, monitoring, and adverse-event costs. Where credible clinical data are available, the costing templates also estimate changes in disease-related costs associated with the use of the new drug. One-way sensitivity analyses, based on variations in the input parameter values, also are included in the more recent costing templates.
An example of an NICE costing template for prasugrel is presented here to illustrate the cost calculator approach for performing a budget-impact analysis. Prasugrel, when coadministered with acetylsalicylic acid, is indicated in the UK for the prevention of atherothrombotic events in patients with acute coronary syndrome (ACS) (that is, unstable angina, nonST segment elevation myocardial infarction (NSTEMI) or ST segment elevation myocardial infarction (STEMI)) who undergo primary or delayed percutaneous coronary intervention. However, prasugrel was recommended by NICE for reimbursement by the NHS as a treatment option for only a subset of the UK-indicated population: those with STEMI, those with STEMI or NSTEMI and stent thrombosis while taking clopidogrel, and those with NSTEMI and diabetes. The NICE costing template is presented in Table 1 and includes the six key elements of a budget-impact analysis estimation of the population size, time horizon (1 year), current and projected treatment mix, drug costs, offsetting disease-related cost savings, and presentation of results. The footnotes to the NICE analysis table provided details of the data sources used in the costing template.
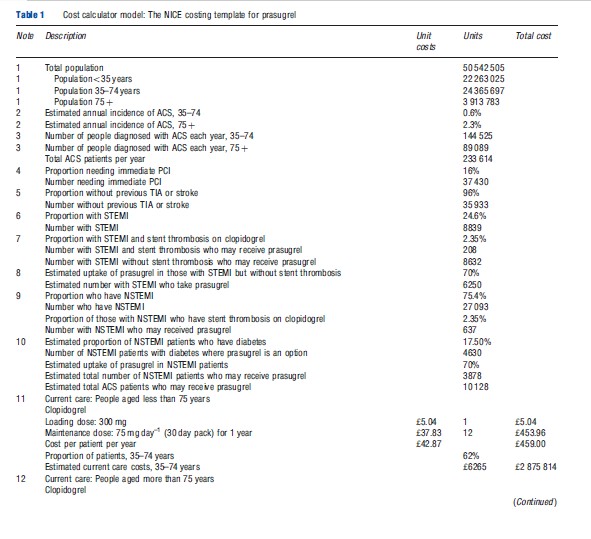
In the prasugrel example, because both drugs included in the analysis are oral drugs, there were no costs estimated for administration. Monitoring costs were also not included. Side effect costs, specifically those associated with bleeding events, were included in the rehospitalization rate. The prasugrel costing template included estimates of savings from a reduced rate of rehospitalization in the first year after the ACS episode that was observed in a large head-to-head clinical trial with clopidogrel.

This NICE costing template for prasugrel also included an extensive one-way sensitivity analysis, using maximum and minimum values for the following input parameter values:
- The annual incidence, by age group.
- The proportion of patients with ACS in whom immediate percutaneous coronary intervention is needed.
- The proportion of patients with STEMI.
- The uptake rate in the STEMI population.
- The proportion of the ACS population with NSTEMI.
- The proportion of NSTEMI population with stent thrombosis on clopidogrel.
- The proportion of the NSTEMI population with diabetes.
- The uptake rate of prasugrel in the NSTEMI population.
- The proportion of patients receiving prasugrel who are more than 75 years of age.
- The cost of clopidogrel per patient per year.
- The reduction in rate of rehospitalizations.
- The weighted average cost of rehospitalizations.
The rationale for the selection of the minimum and maximum values for the sensitivity analysis is not provided in the costing template.
Budget-impact analyses using a cost calculator approach have also been published in peer-reviewed journals. For example, in an article by Chang and Sung, the budget impact of using pimecrolimus cream for atopic dermatitis or eczema was estimated using estimates of the number of people seeking care for this condition each year and the average number of physician visits for the condition each year. Chang and Sung used data from a clinical trial of pimecrolimus to estimate likely reductions in follow-up physician visits for those patients who were treated with pimecrolimus. Although the condition is chronic, it is not progressive or life threatening. Therefore, the use of a static cost calculator approach is appropriate. Chang and Sung estimated the budget impact for a single year, on the basis of observed market share for the first year the drug was introduced, but tested the impact of changes in market uptake in a sensitivity analysis.
Using a static cost calculator approach to budget impact analysis for chronic progressive and/or life-threatening diseases may underestimate the budget impact. For example, Smith and colleagues used a static approach to estimate the budget impact of valsartan for the treatment of patients with heart failure. The authors’ estimates were based on the number of enrollees with heart failure in a US health plan and on the average number of hospitalizations each year for these patients. The authors used data from a clinical trial of valsartan that showed a reduction in the number of hospitalizations and in the length of hospital stay for patients treated with valsartan. However, annual mortality rates with heart failure are significant, and the valsartan clinical trial also estimated a reduction in mortality for patients on valsartan. Such a reduction in mortality would result in an increased number of patients being treated for heart failure at any one time and an associated increase in treatment and monitoring costs for the health plan. This increase in the population size being treated was not included in the Smith and colleagues’ budget-impact analysis. A dynamic disease progression model could have been used to estimate the change in the size of the prevalent population over time, given the reduction in mortality rates. Alternately, estimates of the change in life expectancy with valsartan could have been derived from the clinical trial data and used to estimate the change in the treated population size at steady state and used in the cost calculator approach.
A budget-impact analysis by Dee and colleagues estimated the budget impact of natalizumab over a 3 year time horizon for multiple sclerosis, a slowly progressing chronic disease. In this analysis, the authors explicitly captured the budget impact of the increasing uptake of natalizumab over time. The budget-impact estimates in the Dee and colleagues’ study were based on the reduced costs for treating relapses of multiple sclerosis and the increased drug costs for natalizumab, including administration costs and monitoring for serious side effects such as progressive multifocal leukoencephalopathy. The authors also considered different payer perspectives and adjusted the budget impact depending on which payer perspective was considered. However, this static cost calculator approach ignored the impact on multiple sclerosis treatment costs of slowing the rate of disease progression that is associated with natalizumab treatment.
In the Smith and colleagues’ study, the estimated budget impact of the new treatment did not include the additional drug-related and disease monitoring and symptomatic treatment costs in the extra months of life for the patient. But for patients with heart failure in these studies, the additional life expectancy may be short and the impact on the size of the treated population relatively small. Similarly, the budget impact of slowing disease progression in multiple sclerosis, omitted from the Dee and colleagues’ study, is likely to be small within the time horizon of the budget-impact analysis. But in other chronic conditions, the impact on life expectancy could be significant, for example, for human immunodeficiency virus (HIV) infection. In this case, a dynamic budget-impact model, using either a Markov model or simulation approach, might be more appropriate.
Budget-Impact Analysis: Markov Model Approach
A study by Mauskopf demonstrated how a Markov model can be used to develop both cost-effectiveness and budget-impact estimates for a hypothetical new treatment for HIV infection. To develop the budget-impact estimates, it was first necessary to understand the current distribution of HIV patients among different HIV health states, measured in terms of ranges of CD4 cell counts. This distribution was obtained for a cohort of patients who were not treated, using natural history data that provided estimates of the time spent in each health state. Using these estimates and the number of new patients diagnosed and their CD4 cell-count distribution, the Markov model was run, adding a newly diagnosed cohort each year, until a steady state was reached for the number of patients in each health state without treatment. The introduction of the hypothetical antiretroviral therapy drug regimen was assumed to shift the CD4 cell-count up by one CD4 cell-count range for all patients in the treated cohort and to hold the cohort there for 4 years before disease progression resumed. The Markov model was rerun with the hypothetical antiretroviral drug regimen. For each cycle of the model, the number of individuals alive in each health state was generated. A new steady state was reached in 10–20 years. For each health state, treatment costs, rates of opportunistic infections, and days in the hospital were estimated. Population estimates for all of these outcomes were generated for each year after introduction of the antiretroviral drug regimen.
A Markov budget-impact model can be programed to capture only the budget impact for newly entering cohorts cumulatively in each year after a new drug becomes available; alternatively, the model can be programed to assume that all prevalent patients also immediately switch to the new treatment or that a certain proportion of the prevalent patients switch each year. In the Mauskopf model, there were 10 680 persons alive in the UK with HIV in 1994 and an incident cohort of 1258 persons per year. The treatment regimens compared were no antiretroviral treatment and a hypothetical antiretroviral drug regimen that was assumed to stop disease progression for 4 years but to be taken for 6 years. All persons alive with HIV were assumed to switch immediately to the antiretroviral drug regimen, as were those individuals newly diagnosed during the model time horizon. Selected model inputs and outcomes are shown in Tables 2 and 3, respectively.
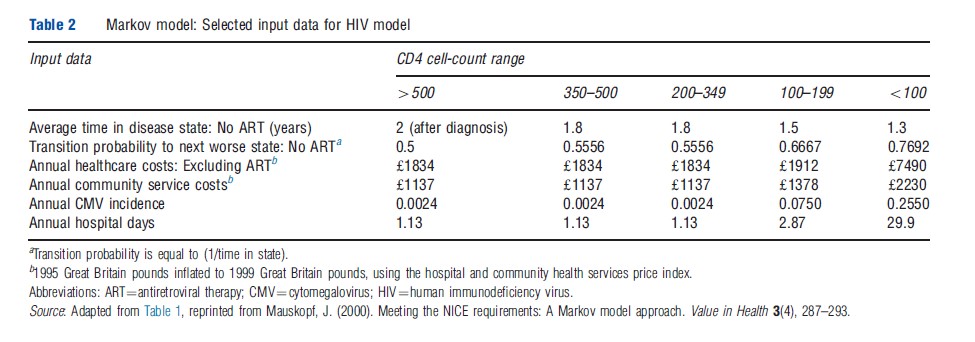
In this model, the impact of antiretroviral therapy (ART) on life expectancy for people with HIV infection was large, resulting in a significant increase in the number of individuals living with acquired immune deficiency syndromes (AIDS) and HIV infection and a shift to less severe disease stages. In this analysis, other outcomes that are of importance to patients and health planners were estimated, including the number of cases of opportunistic infections, illustrated in Table 3 by the number of cases of CMV infection, as well as the number of hospital days used by individuals with HIV infection. This latter value can be very useful for planning for hospital care for those with HIV infection.
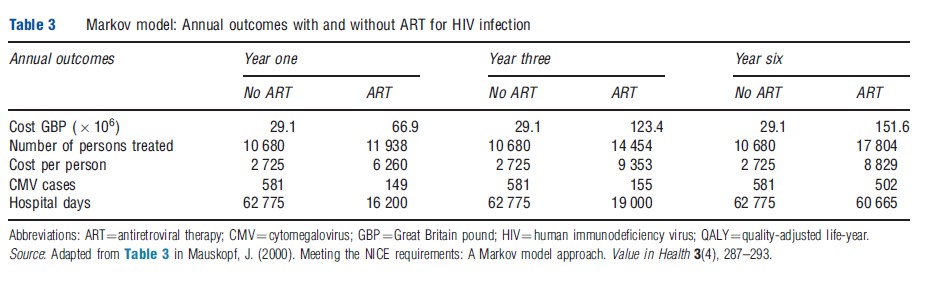
Mar and colleagues presented a similar approach to budget-impact analysis using a Markov model for a Basque population to estimate the impact of the use of thrombolysis for patients with stroke on the prevalence of different degrees of residual disability in patients with stroke and the associated budget impact. In their study, the current prevalent population in different post-stroke health states (death, disability, autonomous, and recurrent stroke) without thrombolysis was estimated using data on stroke incidence stratified by age and sex, all-cause mortality rates, stroke excess mortality risk, and disability outcomes from stroke. The budget impact associated with the use of thrombolysis was estimated using trial data indicating that the percentage of patients with residual disability was lower when thrombolysis was used than when it was not used. Thus, the Markov model was run over a 15 year time horizon with the two different rates of disability, as well as changing numbers of strokes due to the aging population. The results for the Basque population are shown in Table 4. In the Mar’s study, the current population health state prevalence rates, as estimated by the Markov model for patients without thrombolysis, were validated on the basis of population registry data and an alternative modeling approach for estimating poststroke life expectancy.
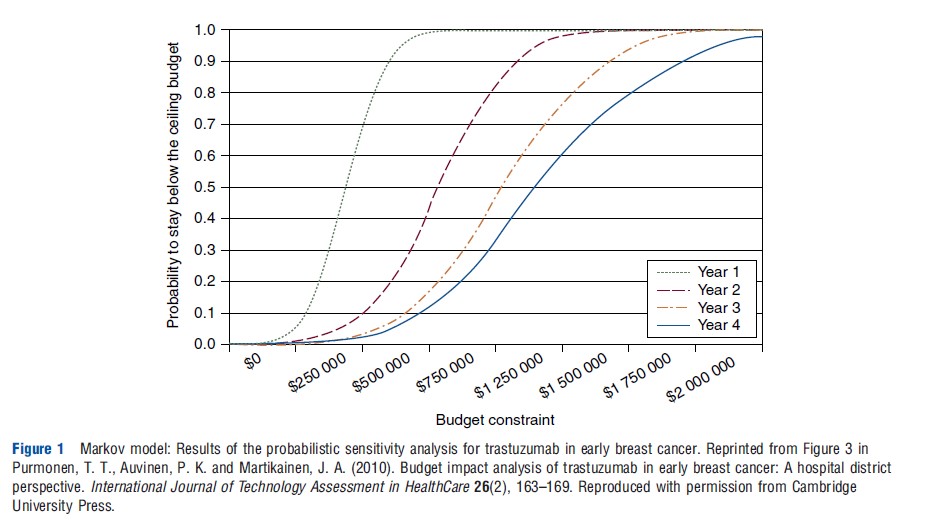
Two other published studies illustrate the use of Markov models to capture the dynamic aspects of budget-impact analysis. In the budget-impact analysis for trastuzumab in early breast cancer by Purmonen and colleagues, a 4 year time horizon was modeled using a state transition model with two health states: free of distant recurrence and with distant recurrence. The budget impact was estimated as the difference in cumulative undiscounted 1, 2, 3, and 4 year costs for all cohorts starting treatment during the model time period with or without the use of adjuvant trastuzumab. The model was based on the number of early breast cancer patients, human epidermal growth factor positive (HER2 þ) prevalence, length and cost of adjuvant treatment, and the effectiveness of the treatment. All HER2 þ patients were assumed to be treated with trastuzumab. Sensitivity analyses included a scenario analysis that looked at different treatment patterns, prevalence of HER2 þ , and treatment costs. In addition, a probabilistic sensitivity analysis was included that estimated the impact of the following uncertain or variable parameter inputs: number of early breast cancer patients, HER2 þ prevalence in those with early breast cancer, disease-related transition probabilities, and treatment costs. The results of the probabilistic sensitivity analysis were presented as an affordability curve in which the probability of the budget impact being below different budget constraints was presented (see Figure 1).
In a combination of cost-utility and budget-impact analysis of third-generation aromatase inhibitors for advanced breast cancer, Marchetti and colleagues used a state transition model to estimate the life expectancy and lifetime costs for a single annual cohort of patients newly diagnosed with advanced breast cancer and starting treatment with or without the use of anastrozole or letrozole. The authors estimated the budget impact for a single cohort under the assumption that all patients in the cohort are treated with either anastrozole or letrozole. This focus on a single cohort and assumption of 100% uptake is typical for cost-effectiveness analyses but is less often used for budget-impact analyses.
Budget-Impact Analysis: Simulation Model Approach
Another type of disease model frequently used in cost-effectiveness analyses of new treatments is Monte Carlo or discrete-event simulation. In simulation models, the disease pathway is simulated for a group of individual patients with different characteristics for the duration of the disease episode or for lifetime (for chronic diseases). This approach to disease modeling has several advantages over a deterministic Markov approach: variability among patients in disease outcomes and in the impact of the treatment is captured explicitly; all relevant patient, system, and treatment characteristics can be captured without requiring an expansion of health states; disease and treatment history over time can be accounted for in the analysis; and multiple events can occur at the same time. Discrete-event simulation models track patients on the basis of the time to the next event, whereas Monte Carlo simulation models typically track the patients at specific time points. The disadvantage of the simulation approach is that it generally requires additional data inputs and additional computation time compared with the Markov modeling approach.
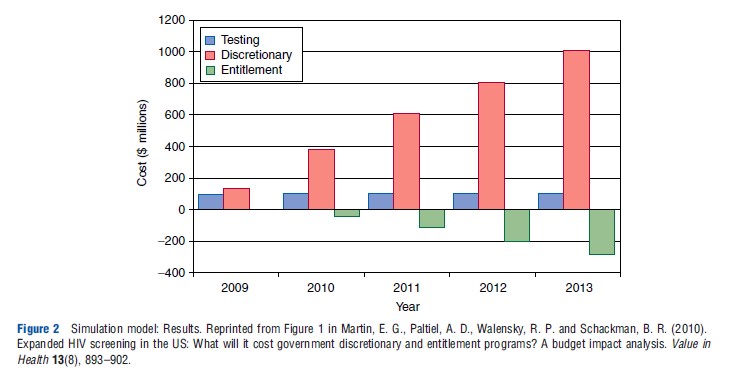
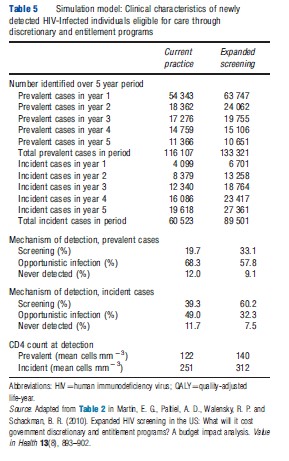
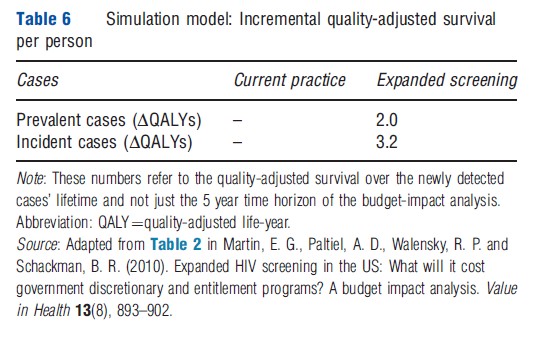
As with Markov modeling, the discrete-event simulation approach can be used to generate budget-impact as well as cost-effectiveness estimates by simulating a prevalent population rather than a single population cohort. Martin and colleagues presented the results of a budget-impact analysis for expanded screening for HIV in the US, using a Monte Carlo simulation model that included screening and treatment for HIV infection. This model has been used extensively for cost-effectiveness analyses of alternative management strategies for HIV infection. In their publication of the simulation model’s results, the authors estimated the number of prevalent cases of HIV infection that were currently undetected and the annual number of new cases of HIV infection, using national prevalence and incidence data. Using a series of published studies and reports, the authors also estimated the proportion of these individuals that would be eligible for government-sponsored HIV screening, as well as the CD4 cell-count and viral load distributions for those persons unaware of their HIV status. The authors then entered this patient population into the screening module of their Monte Carlo simulation model and tracked costs over a 5 year time frame, with and without the introduction of a new screening program. Martin and colleagues presented the additional number of cases identified from expanded screening each year for 5 years and the undiscounted budget impact of expanded screening and the associated earlier treatment by discretionary and entitlement programs (see Tables 5 and 6; Figure 2).
Discrete-event simulation models also have been used to estimate budget impact of drug treatments, tracking both the prevalent and incident populations to determine the annual budget impact. Caro and colleagues used a discrete-event simulation model to estimate the budget impact over 100 days for alternative treatments of bipolar-associated mania, using estimates of changes in response to therapy in the Young Mania Rating Scale over time. Mar and colleagues used a discrete-event simulation model to estimate the budget impact of thrombolysis in patients with stroke, using estimates of a reduced number of patients with residual disability after stroke in those patients given thrombolysis treatment. In both models, prediction equations were estimated by using patient-level data to estimate time to the primary events included in the model.
The advantages of using Monte Carlo or discrete-event simulation models to estimate budget impact of alternative disease management strategies are that, generally such models have been previously validated for the cost-effectiveness analyses and the inputs are consistent for both types of estimates. In addition, changes in disease severity and life expectancy over time can be included in the model. This is very important for HIV infection or stroke, where alternative screening or treatment strategies can have a major impact on the treated population size and/or severity mix, and thus on healthcare decision makers’ budgets.
Conclusions And Where Next
As illustrated in this article, budget-impact models can be developed using a variety of approaches: a cost calculator approach or disease progression modeling approaches using either Markov or simulation models. Generally, the simpler approach is preferred by healthcare decision makers because such an approach is more transparent and can more readily be run using individual health plan characteristics. The cost calculator approach can be used for acute illnesses, as well as for chronic illness where changes in disease severity, life expectancy, or treatment patterns (1) do not occur, (2) occur very rapidly and can readily be captured in a cost-calculator model, or (3) occur beyond the time horizon of the budgetimpact analysis. In instances where the changes in disease severity, life expectancy, and/or treatment patterns cannot be credibly captured in a cost-calculator model, a disease progression modeling approach might be needed.
A disease progression modeling approach may be more desirable when an integrated cost-effectiveness and budgetimpact model is desired. However, care needs to be taken to ensure that the budget-impact estimates are generated for the prevalent population rather than for the single-disease cohort that is typically used for cost-effectiveness analysis. The budget-impact model should also compare a mix of current and future treatments rather than a simple comparison of all patients treated with either a current treatment or a new treatment, as is typically seen in a cost-effectiveness analysis. In addition, the appropriate costs for the budget holder are their actual prices paid net of discounts and copays while opportunity costs are more appropriately used for cost-effectiveness analyses.
The question of how to reflect the uncertainty or variability in the inputs to a budget-impact analysis is also important. There are several different types of uncertainty or variability that can be present in the input parameter values, uncertainty about the estimates of the efficacy of the new and current interventions, variability in patient characteristics and current treatment patterns in different healthcare settings, and both uncertainty and variability in the changes in expected treatment patterns with the availability of the new intervention. Because these analyses are aimed to help healthcare decision makers understand the budget impact on the population for which the decision makers have responsibility, budget-impact analyses most commonly include either one-way sensitivity analyses, using ranges of both uncertain efficacy inputs and differences in patient characteristics and current and future treatment patterns (e.g., NICE cost calculators), or scenario analyses where several of these input parameter values may be changed to produce a scenario that most closely matches the healthcare decision maker’s population. Probabilistic sensitivity analyses are sometimes included in published budgetimpact analyses, but these are probably not very useful for healthcare decision makers because the sensitivity of the budget-impact analyses results to parameter uncertainty may be less than the sensitivity of the budget-impact analysis to variabilities in the healthcare decision maker’s population characteristics and treatment patterns. The concept of the affordability curve for different budget constraints as used in Purmonen and colleagues’ article may be a useful way to present the results of a probabilistic sensitivity analysis. However, the probabilistic sensitivity analysis presented in Purmonen and colleagues’ study included both uncertain parameters (HER2 þ prevalence and transition probabilities reflecting efficacy) and variable parameters that would probably be known with certainty by the decision maker (price of trastuzumab and number of patients), thus reducing the value of their probabilistic sensitivity analysis.
Although the primary purpose of a budget-impact model is to estimate the annual impact on a health plan budget after a new intervention is reimbursed for the health plan’s covered population, a budget-impact models may also generate estimates of the associated changes in population health outcomes during the same time period. These estimates may be used in the budget-impact analysis to estimate the changes in disease-related costs, but the population health estimates also can provide useful information for healthcare decision makers. For example, estimates of changes in disease cases or hospital days may be useful for health services planners. These population-based estimates of these outcome changes should be presented for each year after introduction of the new intervention along with the budget-impact estimates.
Bibliography:
- Caro, J. J., Huybrechts, K. F., Xenakis, J. G., et al. (2006). Budgetary impact of treating acute bipolar mania in hospitalized patients with quetiapine: An economic analysis of clinical trials. Current Medical Research and Opinion 22, 2233–2242.
- Chang, J. and Sung, J. (2005). Health plan budget impact analysis for pimecrolimus. Journal of Managed Care Pharmacy 11, 66–73.
- Danese, M. D., Reyes, C., Northridge, K., et al. (2008). Budget impact model of adding erlotinib to a regimen of gemcitabine for the treatment of locally advanced, nonresectable or metastatic pancreatic cancer. Clinical Therapeutics 30, 775–784.
- Dasbach, E. J., Largeron, N. and Elbasha, E. H. (2008). Assessment of the cost-effectiveness of a quadrivalent HPV vaccine in Norway using a dynamic transmission model. Expert Review of Pharmacoeconomics and Outcomes Research 8, 491–500.
- Dee, A., Hutchinson, M. and De La Harpe, D. (2012). A budget impact analysis of natalizumab use in Ireland. Irish Journal of Medical Sciences 181, 199–204.
- Mar, J., Arrospide, A. and Comas, M. (2010). Budget impact analysis of thrombolysis for stroke in Spain: A discrete event simulation model. Value in Health 13, 69–76.
- Mar, J., Sainz-Ezkerra, M. and Miranda-Serrano, E. (2008). Calculation of prevalence with Markov models: Budget impact analysis of thrombolysis for stroke. Medical Decision Making 28, 481–490.
- Marchetti, M., Caruggi, M. and Colombo, G. (2004). Cost utility and budget impact of third-generation aromatase inhibitors for advanced breast cancer: A literature based model analysis of costs in the Italian National Health Service. Clinical Therapeutics 26, 1546–1561.
- Martin, E. G., Paltiel, A. D., Walensky, R. P. and Schackman, B. R. (2010). Expanded HIV screening in the U.S.: what will it cost government discretionary and entitlement programs? A budget impact analysis. Value in Health 13, 893–902.
- Mauskopf, J. (2000). Meeting the NICE requirements: A Markov model approach. Value in Health 3, 287–293.
- Mauskopf, J., Murroff, M., Gibson, P. J. and Grainger, D. L. (2002). Estimating the costs and benefits of new drug therapies: Atypical antipsychotic drugs for schizophrenia. Schizophrenia Bulletin 28, 619–635.
- Purmonen, T. T., Auvinen, P. K. and Martikainen, J. A. (2010). Budget impact analysis of trastuzumab in early breast cancer: A hospital district perspective. International Journal of Technology Assessment in Health Care 26, 163–169.
- Smith, D. G., Cerulli, A. and Frech, F. H. (2005). Use of valsartan for the treatment of heart-failure patients not receiving ACE inhibitors: A budget impact analysis. Clinical Therapeutics 27, 951–959.
- Sullivan, S. D., Mauskopf, J. A., Augustovski, F., et al. (forthcoming). Budget Impact Analysis – Principles of Good Practice: Report of the ISPOR 2012 Budget Impact Analysis Good Practice II Task Force. Value Health.
- Wiviott, S. D., Braunwald, E., McCabe, C. H., et al. TRITON-TIMI 38 Investigators (2007). Prasugrel versus clopidogrel in patients with acute coronary syndromes. New England Journal of Medicine 357, 2001–2015.